Introduction
In biostatistics, researchers frequently compare more than two groups to understand biological, medical, or environmental differences. Whether evaluating drug efficacy across treatment groups, comparing physiological parameters among species, or analyzing clinical trial outcomes, Analysis of Variance (ANOVA) is commonly used.
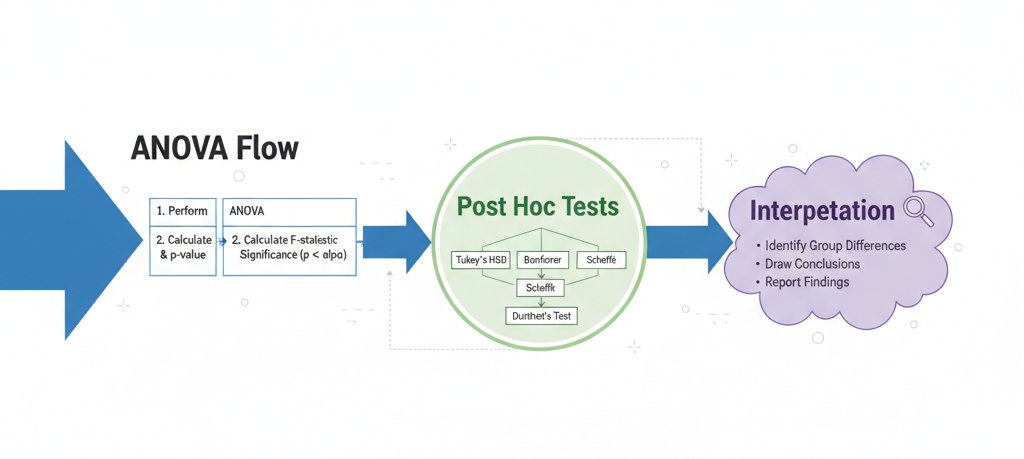
However, a statistically significant ANOVA result only tells us that at least one group differs, not which specific groups differ from each other. This is where post hoc multiple comparison tests play a crucial role.
Post hoc tests allow researchers to make pairwise comparisons while controlling for Type I error, ensuring that conclusions remain statistically valid. In biostatistics—where decisions can impact health, medicine, and ecological management—choosing the correct post hoc test is essential.
Why Are Post Hoc Tests Necessary in Biostatistics?
When multiple groups are compared, the probability of making a false positive (Type I error) increases with each additional comparison.
For example:
- Comparing 4 treatment groups involves 6 pairwise comparisons
- Each comparison increases the chance of incorrectly declaring significance
Post hoc tests:
- Control the family-wise error rate (FWER)
- Identify specific group differences
- Improve scientific accuracy and reproducibility
In biostatistical research, incorrect conclusions can lead to:
- Misinterpretation of treatment effects
- Invalid clinical recommendations
- Flawed biological inference
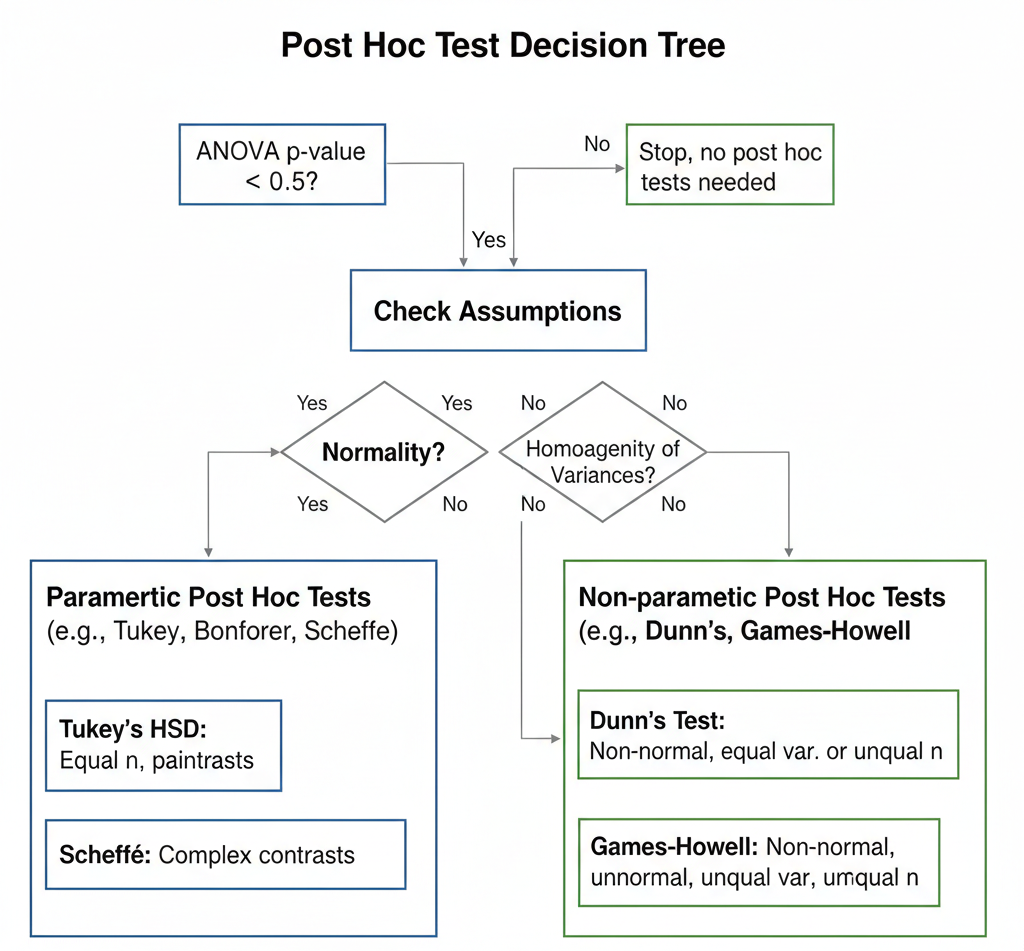
When Should Post Hoc Multiple Comparison Tests Be Applied?
Post hoc tests are applied when:
- ANOVA is statistically significant (p < 0.05)
- The study involves three or more independent groups
- The researcher needs to identify which groups differ
- Assumptions of ANOVA are reasonably met (normality, homogeneity)
⚠️ Important:
Post hoc tests should not be used if ANOVA is non-significant.
Commonly Used Post Hoc Tests in Biostatistics
Different post hoc tests are designed for different data structures and research goals. Below are the most widely used tests in biological and medical sciences.
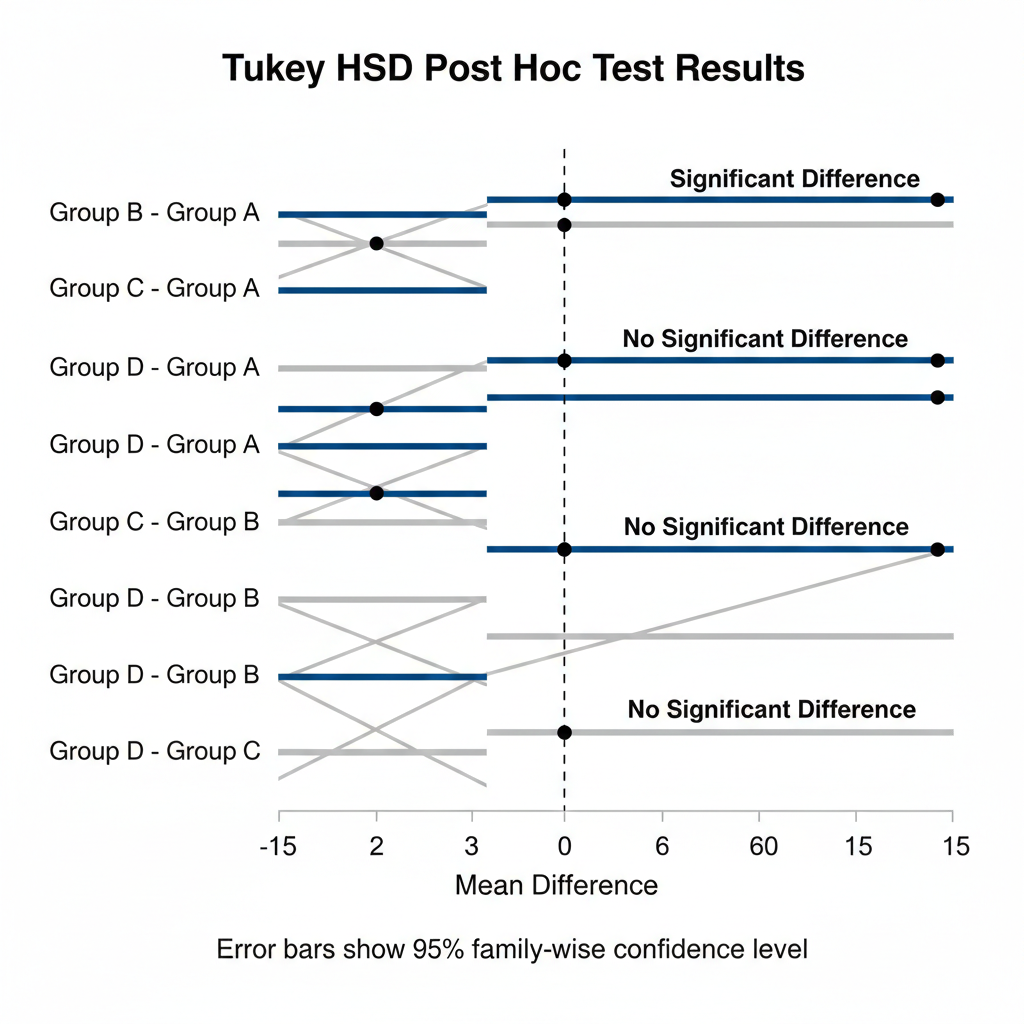
1. Tukey’s Honestly Significant Difference (HSD) Test
Tukey’s HSD is one of the most popular post hoc tests in biostatistics.
Key features:
- Compares all possible pairwise means
- Controls family-wise error rate
- Best suited for equal sample sizes
Applications in biostatistics:
- Comparing enzyme activity across treatments
- Analyzing plant growth under multiple fertilizers
- Evaluating laboratory animal responses
Advantages:
- Strong error control
- Easy interpretation
- Widely accepted by journals
Limitations:
- Less powerful with unequal sample sizes
2. Bonferroni Correction
Bonferroni is a conservative adjustment method rather than a true test.
How it works:
- Divides α (e.g., 0.05) by number of comparisons
- Example: α / 6 = 0.0083
Applications:
- Clinical trials
- Studies where false positives must be minimized
Advantages:
- Very strict error control
- Simple calculation
Limitations:
- Low statistical power
- Increased risk of Type II error
3. Scheffé’s Test
Scheffé’s test is extremely flexible and conservative.
Key features:
- Suitable for unequal sample sizes
- Allows complex comparisons, not just pairwise
Applications:
- Ecological studies
- Observational biological research
- Retrospective data analysis
Advantages:
- Strong control of Type I error
- Very flexible
Limitations:
- Low sensitivity
- May miss true differences
4. Dunnett’s Test
Dunnett’s test is used when all groups are compared to a single control group.
Applications:
- Drug vs placebo studies
- Toxicological dose-response studies
- Biomedical experiments
Advantages:
- More powerful than Tukey for control-based comparisons
- Reduces unnecessary comparisons
Limitations:
- Cannot compare treatment groups with each other
5. Least Significant Difference (LSD) Test
LSD is one of the simplest post hoc tests.
Key features:
- No correction for multiple testing
- High sensitivity
Applications:
- Exploratory studies
- Pilot biological research
Limitations:
- High risk of false positives
- Not recommended for confirmatory studies
Non-Parametric Post Hoc Tests in Biostatistics
When data violate ANOVA assumptions, non-parametric post hoc tests are preferred.
Common options:
- Dunn’s Test (after Kruskal–Wallis test)
- Nemenyi Test
- Pairwise Wilcoxon with adjustment
These tests are commonly used in:
- Microbial count data
- Ecological abundance data
- Ordinal clinical scores
Comparison Table of Post Hoc Multiple Comparison Tests
| Test Name | Best Used When | Controls Type I Error | Equal Sample Size Required | Common Applications |
|---|---|---|---|---|
| Tukey HSD | All pairwise comparisons | Yes | Yes (preferred) | Biology, ecology |
| Bonferroni | Few comparisons | Yes (very strict) | No | Clinical research |
| Scheffé | Complex contrasts | Yes | No | Observational studies |
| Dunnett | Compare with control | Yes | No | Toxicology, medicine |
| LSD | Exploratory analysis | No | Yes | Pilot studies |
| Dunn’s Test | Non-parametric data | Yes | No | Microbiology |
How to Choose the Right Post Hoc Test in Biostatistics
Choosing the correct test depends on:
- Study design
- Number of groups
- Presence of a control group
- Sample size balance
- Distribution of data
General guidelines:
- Use Tukey HSD for balanced biological experiments
- Use Dunnett’s test for treatment vs control
- Use Scheffé’s test for flexible comparisons
- Use Dunn’s test for non-normal data
Common Mistakes to Avoid
- Applying post hoc tests without significant ANOVA
- Using LSD for confirmatory studies
- Ignoring unequal sample sizes
- Not reporting adjusted p-values
- Overinterpreting marginal significance

How to Report Post Hoc Results in Biostatistics
A proper scientific report should include:
- Name of post hoc test used
- Adjustment method
- Mean differences
- Confidence intervals
- Adjusted p-values
Example statement:
“Post hoc analysis using Tukey’s HSD revealed that Treatment A differed significantly from Treatment C (p = 0.021), while no significant difference was observed between Treatments A and B.”
Conclusion
Post hoc multiple comparison tests are an essential component of biostatistical analysis. They bridge the gap between a significant ANOVA result and meaningful biological interpretation. By controlling Type I error and identifying specific group differences, post hoc tests ensure scientific rigor and reproducibility.
In biostatistics, where outcomes influence medical, ecological, and biological decisions, selecting the appropriate post hoc test is critical. Understanding their assumptions, strengths, and limitations allows researchers to make valid and defensible conclusions.
When applied correctly, post hoc multiple comparison tests transform raw statistical output into clear biological insight.