Introduction
Biological risk assessment is one of the most important components of modern biostatistics, public health, biotechnology, toxicology, pharmacology, environmental sciences, and epidemiology. It involves quantifying the probability that a biological agent, process, exposure, or hazard will cause an adverse effect on human health, animals, or the environment.
Biostatistics provides the mathematical and analytical foundation to measure, compare, and predict biological risks using reliable data. Whether assessing the toxicity of a chemical, the infection risk of a pathogen, the carcinogenicity of pollutants, or the adverse effects of drugs, biostatistical tools such as dose–response models, logistic regression, survival analysis, confidence intervals, and probability estimation play a central role.
What Is Biological Risk Assessment?
Biological risk assessment is the process of identifying, analyzing, and evaluating biological hazards to estimate the likelihood and severity of potential harm.
Key components include:
- Hazard Identification: What biological agent or exposure can cause harm?
- Dose–Response Assessment: At what level does exposure cause an effect?
- Exposure Assessment: How much exposure occurs in real-world settings?
- Risk Characterization: Integration of all data to quantify risk.
Biostatistics ensures that each step relies on evidence-based methods, not assumptions.
Importance of Biostatistics in Biological Risk Assessment
Biostatistics plays the following roles:
1. Data Organization and Description
- Summary statistics (mean, SD, median, percentiles)
- Data visualization (histograms, boxplots, scatter plots)
2. Estimation of Risk Levels
- Probability of disease occurrence
- Estimation of lethal dose (LD50)
- Infection risk modeling
- Hazard ratios (HR)
3. Hypothesis Testing
- Comparing exposure groups
- Testing biological differences
- Identifying statistically significant risk factors
4. Predictive Modeling
- Logistic regression
- Probit analysis
- Cox proportional hazards model
- Bayesian risk models
5. Uncertainty and Sensitivity Analysis
- Confidence intervals
- Bootstrapping
- Monte Carlo simulations
Core Components of Biological Risk Assessment

1. Hazard Identification
This step describes the biological agent or exposure. Examples include:
- Pathogenic bacteria (e.g., Salmonella, E. coli)
- Viruses (e.g., Influenza, SARS-CoV-2)
- Chemical contaminants
- Toxins
- Pharmaceuticals
- Pesticides
- Radiation exposure
- Genetically modified organisms (GMOs)
Statistical tools used:
- Evidence mapping
- Descriptive statistics
- Systematic review methods
2. Exposure Assessment
Biostatistics helps estimate how much, how long, and how often exposure occurs.
Common Statistical Techniques
- Time-series analysis
- Dose calculations
- Probability distributions
- Exposure frequency tables
- Environmental sampling statistics
3. Dose–Response Assessment
This is one of the most statistical steps in the entire process.
The goal is to determine the quantitative relationship between exposure level and biological response.
Biostatistical Models Used
- Probit regression
- Logit models (Logistic regression)
- Weibull models
- Hill equation models
- Linear and nonlinear regression models
Common Metrics
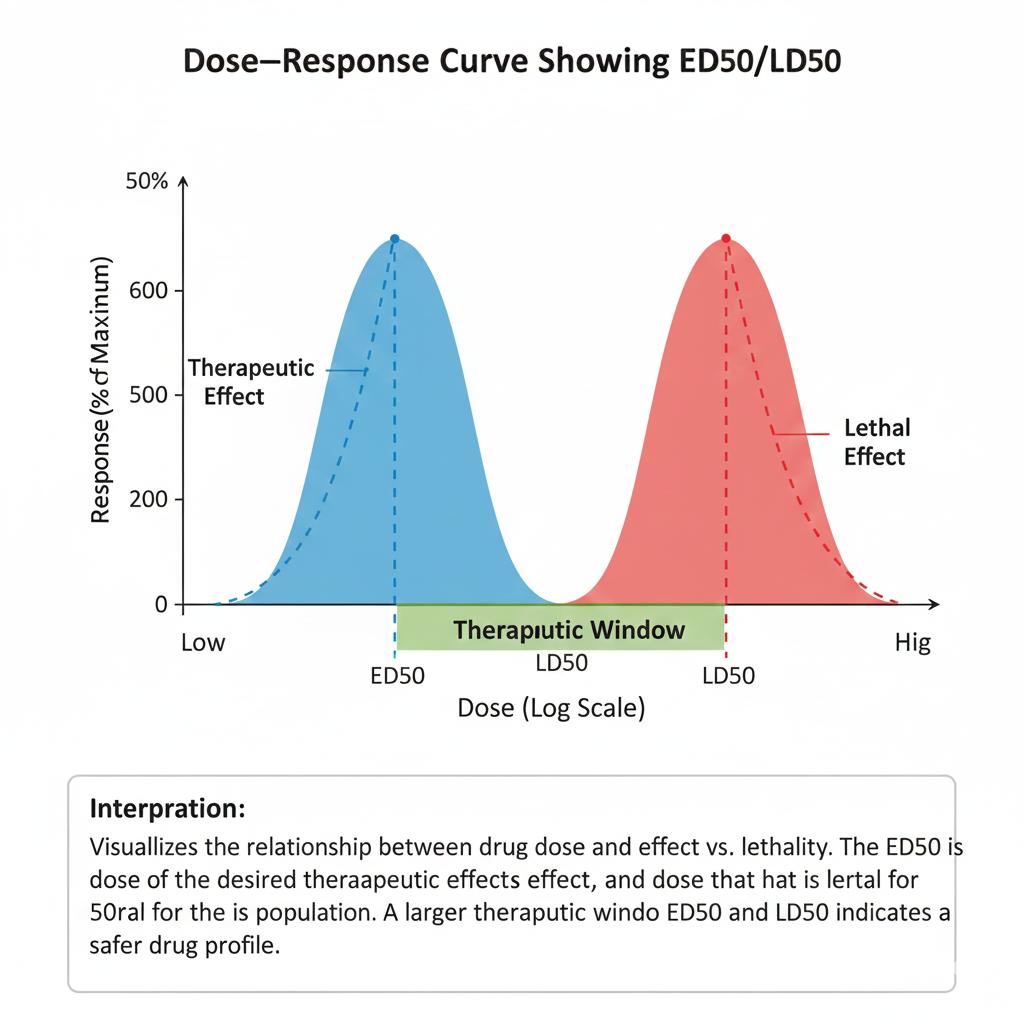
- LD50 (Lethal Dose 50%): dose where 50% of population dies
- ED50 (Effective Dose 50%): dose where 50% shows response
- NOAEL (No-Observed-Adverse-Effect Level)
- LOAEL (Lowest-Observed-Adverse-Effect Level)
These values are estimated using biostatistical probability models.
4. Risk Characterization
This integrates data from previous steps and calculates:
- Overall probability of harm
- Severity estimates
- Uncertainty margins
- Population variability
- Confidence intervals
Outcome presentation may include:
- Risk curves
- Probability tables
- Hazard ratio plots
- Monte Carlo simulation outputs
Statistical Methods Commonly Used in Biological Risk Assessment
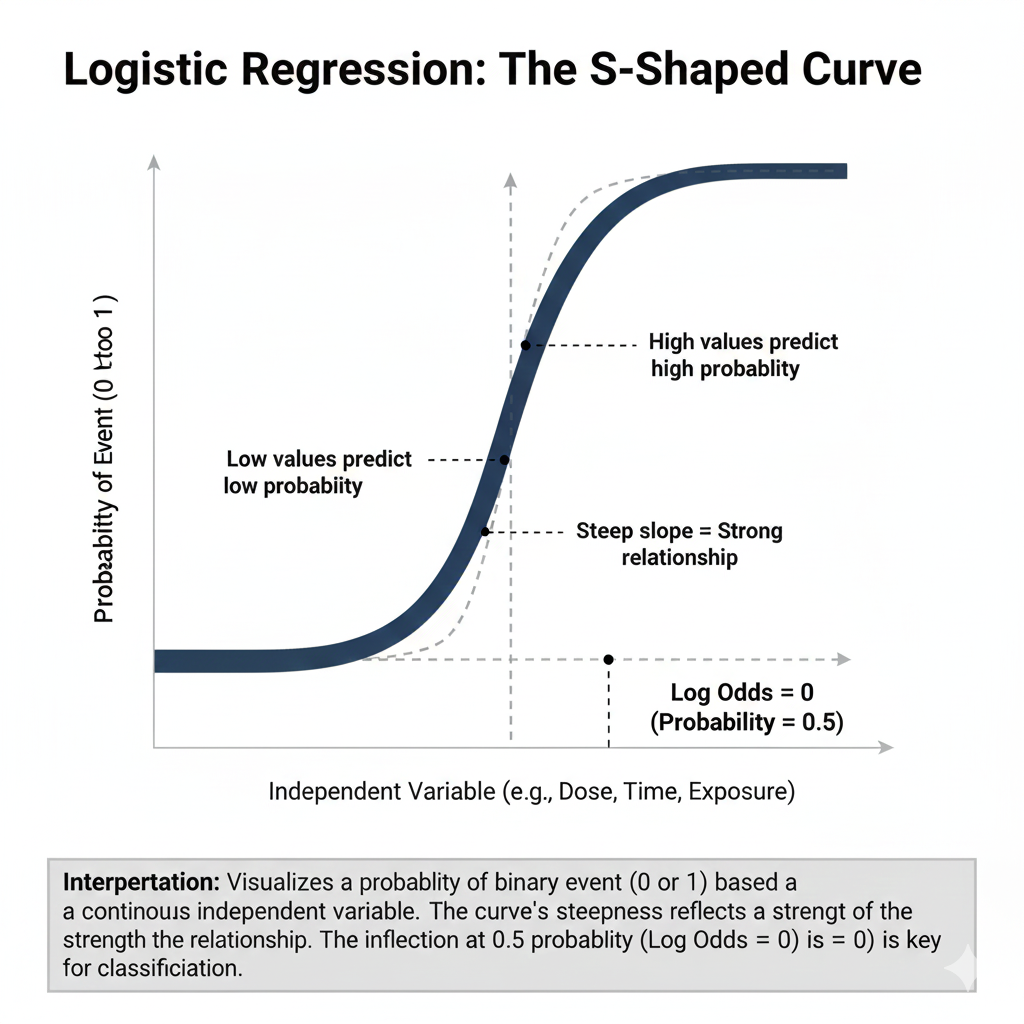
1. Logistic Regression
Used when response is binary:
- Disease vs. No disease
- Alive vs. Dead
- Toxic vs. Non-toxic
Formula
P(Y=1) = 1 / 1+e−(β0+β1X)
Interpretation
- Odds ratio (OR) quantifies risk
- OR > 1 = Increased risk
- OR < 1 = Protective factor
2. Probit Analysis
Used for toxicology studies.
Probit(p) = β0 + β1 log(Dose)
Useful for estimating:
- LD50
- ED50
- Toxic concentrations
3. Cox Proportional Hazards Model (Survival Analysis)
Used for long-term biological effects.
h(t) = h0(t)e(β1X1+β2X2+…)
Outputs include:
- Hazard ratio (HR)
- Survival curves
- Time-dependent risks
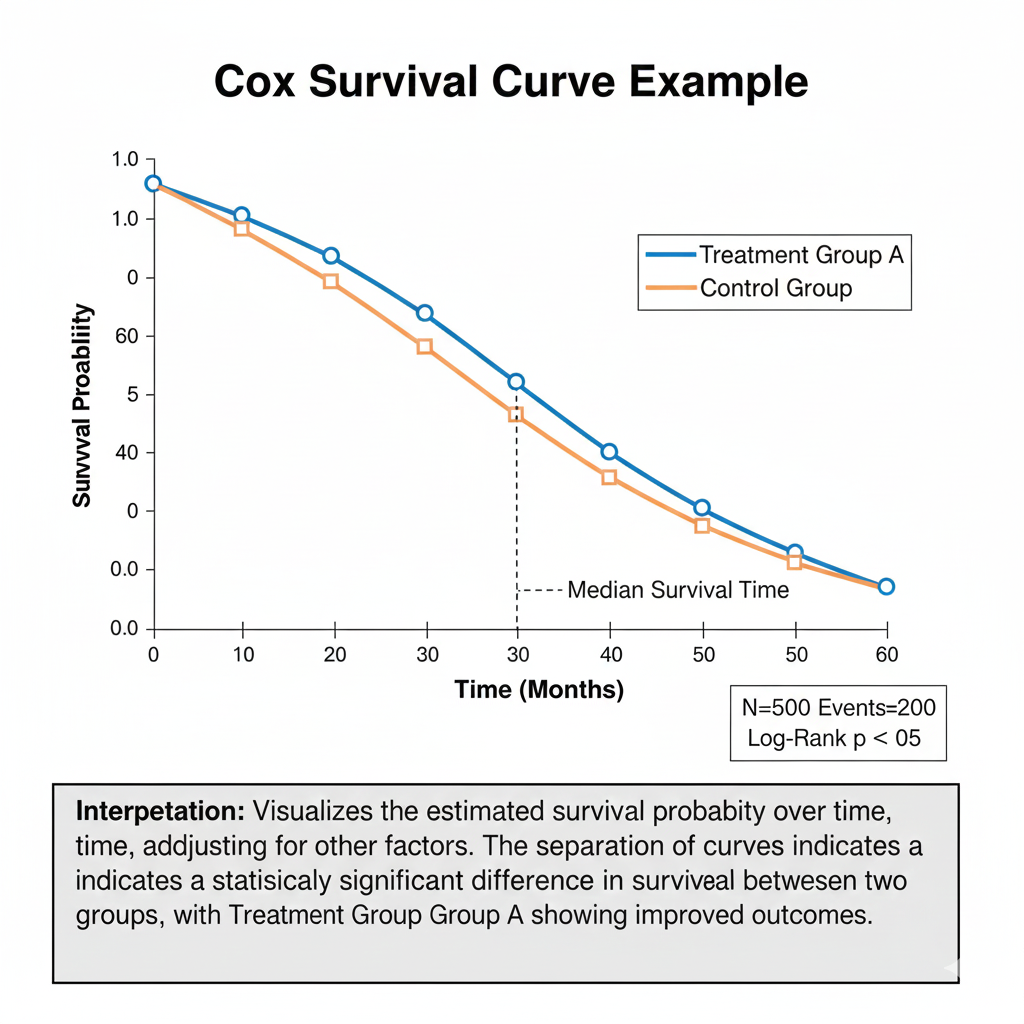
Table: Overview of Biological Risk Assessment Steps and Biostatistical Tools
| Model | Application in Risk Assessment | Key Output |
|---|---|---|
| Logistic Regression | Binary risk prediction | Odds Ratio |
| Probit Model | Toxicology dose–response | LD50, ED50 |
| Cox Model | Time-to-event risk | Hazard Ratio |
| Linear Regression | Continuous exposure outcomes | Slope, CI |
| Bayesian Models | Uncertainty estimation | Posterior risk |
Table: Common Biostatistical Models and Their Applications
| Model | Application in Risk Assessment | Key Output |
|---|---|---|
| Logistic Regression | Binary risk prediction | Odds Ratio |
| Probit Model | Toxicology dose–response | LD50, ED50 |
| Cox Model | Time-to-event risk | Hazard Ratio |
| Linear Regression | Continuous exposure outcomes | Slope, CI |
| Bayesian Models | Uncertainty estimation | Posterior risk |
Applications of Biological Risk Assessment
1. Public Health and Epidemiology
- Predicting infection spread
- Identifying high-risk populations
- Vaccine response analysis
- Outbreak modeling
2. Environmental Risk Assessment
- Water quality toxicity
- Soil contamination
- Ecosystem exposure risks
- Airborne pollutants
3. Pharmaceutical and Clinical Research
- Clinical trial adverse effects
- Dose-finding studies
- Safety margin estimation
- Therapeutic window calculation
4. Food Safety and Agriculture
- Pesticide toxicity
- Microbial contamination risks
- GM crop safety evaluation
Factors Affecting Biological Risk
Biostatistical models consider variables such as:
- Age
- Gender
- Genetics
- Immune status
- Environmental exposure
- Nutritional factors
- Duration of exposure
- Dose level
These are typically included as covariates in models.
Example Scenario: Estimating LD50 in a Toxicology Experiment
Suppose different doses of a chemical were given to laboratory animals. Using probit analysis, biostatisticians calculate:
- Probability of death at each dose
- LD50 with 95% CI
- Slope of dose–response curve
- Goodness of fit (χ²)
This helps determine whether the substance is safe at low doses.
Conclusion
Biological risk assessment is a vital process in biostatistics, bridging quantitative data with biological understanding to evaluate risks accurately. Biostatistics provides the essential tools required to estimate exposure levels, model dose–response relationships, and predict health outcomes with precision.
From logistic regression and probit models to survival analysis and Monte Carlo simulations, the techniques used in biological risk assessment allow researchers to understand, quantify, and communicate risks. These assessments guide public health policies, environmental safety decisions, pharmaceutical dosages, and disease prevention strategies.
A proper biological risk assessment ensures safety, minimizes harm, and supports evidence-based decision making in biological and medical sciences.